
Basic Standards for
Home/Commercial Kitchens
and Small Scale Food
Processing Facilities
Prepared By:
NYS Department of Agriculture and Markets
Division of Food Safety and Inspection

Agenda
Regulations
Zoning/Jusidication - Licensing and exemptions
Specialized processing (HACCP)
Acidified foods, Juice, Seafood, Low acid canned foods
Overview of CURRENT GMPs
Labeling
Allergens
Food recalls
Employee training opportunities
Scheduled processes/authorities
FSMA
Resources

Regulations – Why do we have them?
The Federal government, individual states, cities and municipalities govern the operation of
food processing facilities whether home kitchens or commercial facilities.
When considering starting up a home or commercial kitchen, it is important to research the
following:
Which agencies regulates/licenses your product,
Foods allowed and not allowed to be produced in each facility,
Local zoning laws governing the use of the building
Foods that are regulated and require a Article 20-C Food Processing License in New York
This regulation applies to anything that is altered by value-added processing including
but not limited to: baking, blending, brewing, curing, fermenting, freezing, grinding,
pickling, canning, preserving, dehydrating, juicing, cider making, pickling, brining,
bottling, packaging, repackaging, vacuum or reduced oxygen packaging, pressing,
heating or cooking, smoking, roasting, or manufacturing.
Complete list in handout
Zoning Regulations? – Jurisdiction?
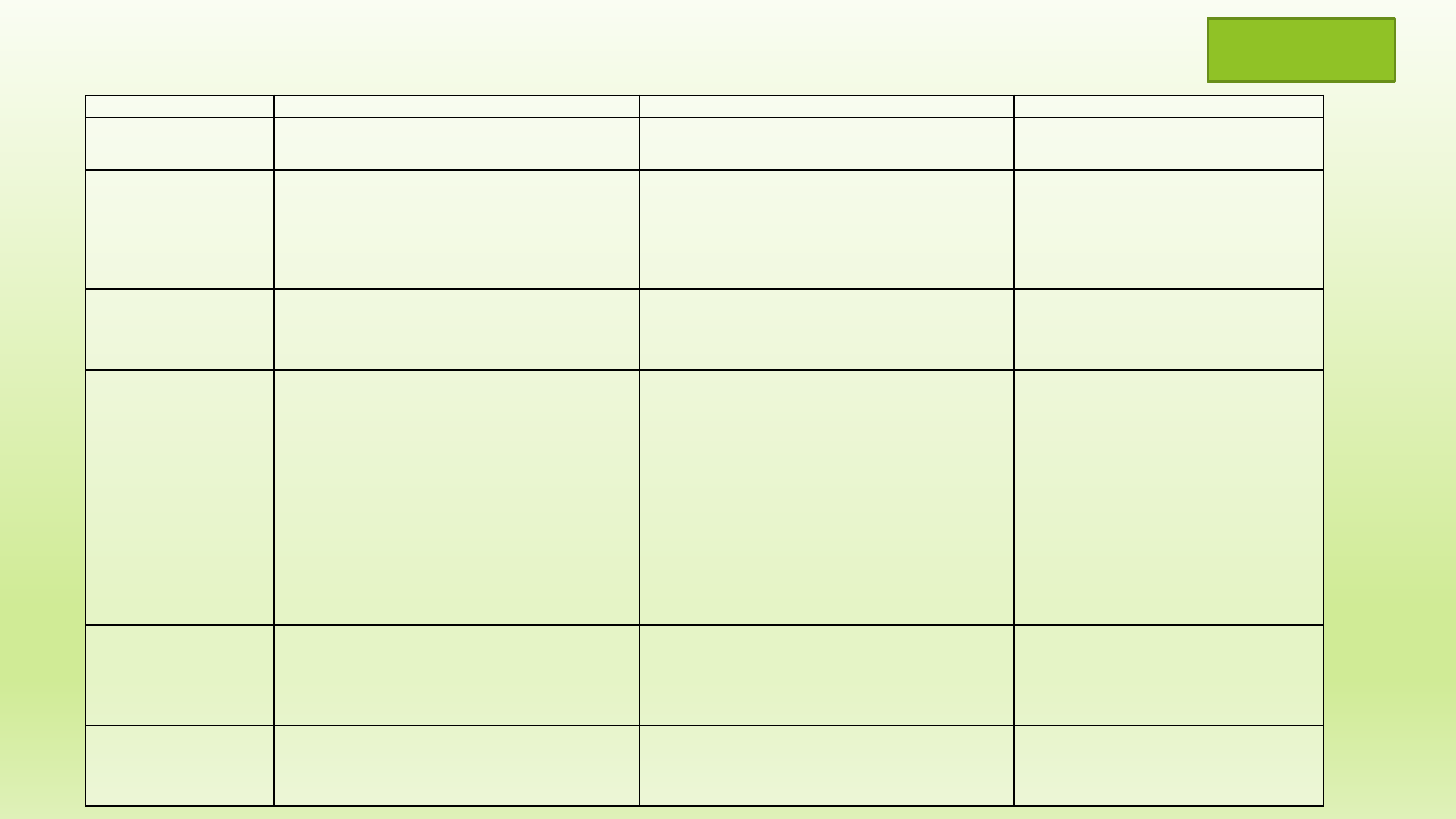
Procedure
Home Kitchen Home Annex (if licensable) Commercial
Inspection
No, Potable water required (documented)
–
municipal or treated well water
Yes, Potable water required (documented)
–
municipal or treated well water
Yes, Potable water required (documented) –
municipal or treated well water
Licensing
Non
-potentially hazardous foods exempt from
licensing by
NYS Department of Agriculture &
Markets (NYSDAM)
Wholesale or retail is allowed at agricultural venues
(farmers markets, craft fairs, etc.)
20
-C license
NYS Dept. of Agriculture &Markets
Separate cleaning, sanitizing, and hand wash
facilities.
Easily cleanable walls, floor, and ceiling.
Fee: $400.00/2 years
20
-C license
NYSDAM
Fee
– $400.00/2 years
Inspection Agency
NYSDAM
May request review of processing procedures by
recognized processing authority. Only normal
kitchen facilities can be used.
NYSDAM
NYSDAM
Food service facilities are inspected by the
Department of Health.
Foods Allowed
Candy
- non-chocolate, fudge
Cakes not requiring refrigeration
Cookies
Brownies
Double crust
fruit pies
Breads
(Not containing Fruit/Vegetables)
Rolls
Fruit jams, jellies
Spices, herbs (blending commercially dried)
Snack items
Baked goods (i.e. bread, rolls) for wholesale
distribution or retail at farmers markets, craft fairs,
etc.
Any processed food
Process Review required for any shelf stable foods.
May be required for other foods as well (i.e.
fermented)
Scheduled Process is required for Low acid and
acidified foods packed in hermetically sealed
containers.
Must register and file with FDA
Any processed food
Process Review required for any shelf stable
foods. May be required for other foods as
well (i.e. fermented)
Scheduled Process is required for Low acid
and acidified foods packed in hermetically
sealed containers.
Must register and file
with
FDA
Foods Not Allowed
Cakes which require refrigeration
Pies containing milk, eggs or meat products
Chocolates
Low acid / acidified foods
Etc.
Meat products
– if more than 3% raw or 2% cooked
meat ingredients
– USDA regulated
Meat products
if more than 3% raw or 2%
cooked meat ingredients
– USDA regulated
Zoning
Check with city/town Zoning /Planning Board
Issues: Scale of operation, number of employees
Check with municipality Zoning/Planning Board
Issues: scale of operation; number of employees
2nd kitchen may not be allowed on premise
Check with municipality Zoning/Planning
Board
Issues: scale of operation, number of
employees
Covered in
handout!

Home Processing Exemption
New York State allows non-hazardous foods such as candy, cakes not requiring refrigeration, cookies,
brownies, double crusted fruit pies, breads and rolls, standard fruit jams and jellies, repackaging
commercially dried spices and herbs, and snack items to be produced in home kitchens.
A review of processing procedures may be required for certain products before exemption is granted.
Anyone seeking a Home Processing Exemption must contact the NYS Department of Agriculture &
Markets. Home Processor information can be found on our website:
https://www.agriculture.ny.gov/FS/consumer/FSI-898D_Home_Processor.pdf Any questions can be
emailed to agr.sm.hpregistrations@agriculture.ny.gov
NOTE: An annual potable water test for bacteria is required for all home processors on private water
supplies. Internet sales are not allowed under this exemption.
Some types of foods may not be produced in a home kitchen, as mandated by Federal regulations.
These foods include :
Low acid and acidified (pickled) foods packed in hermetically sealed containers must be registered
with the US Food and Drug Administration (FDA).
Meat products with more than 3% raw or 2% cooked meat ingredients in a completed product are
regulated by the US Department of Agriculture (USDA).
Vacuum packaged and any other reduced oxygen packaged products.
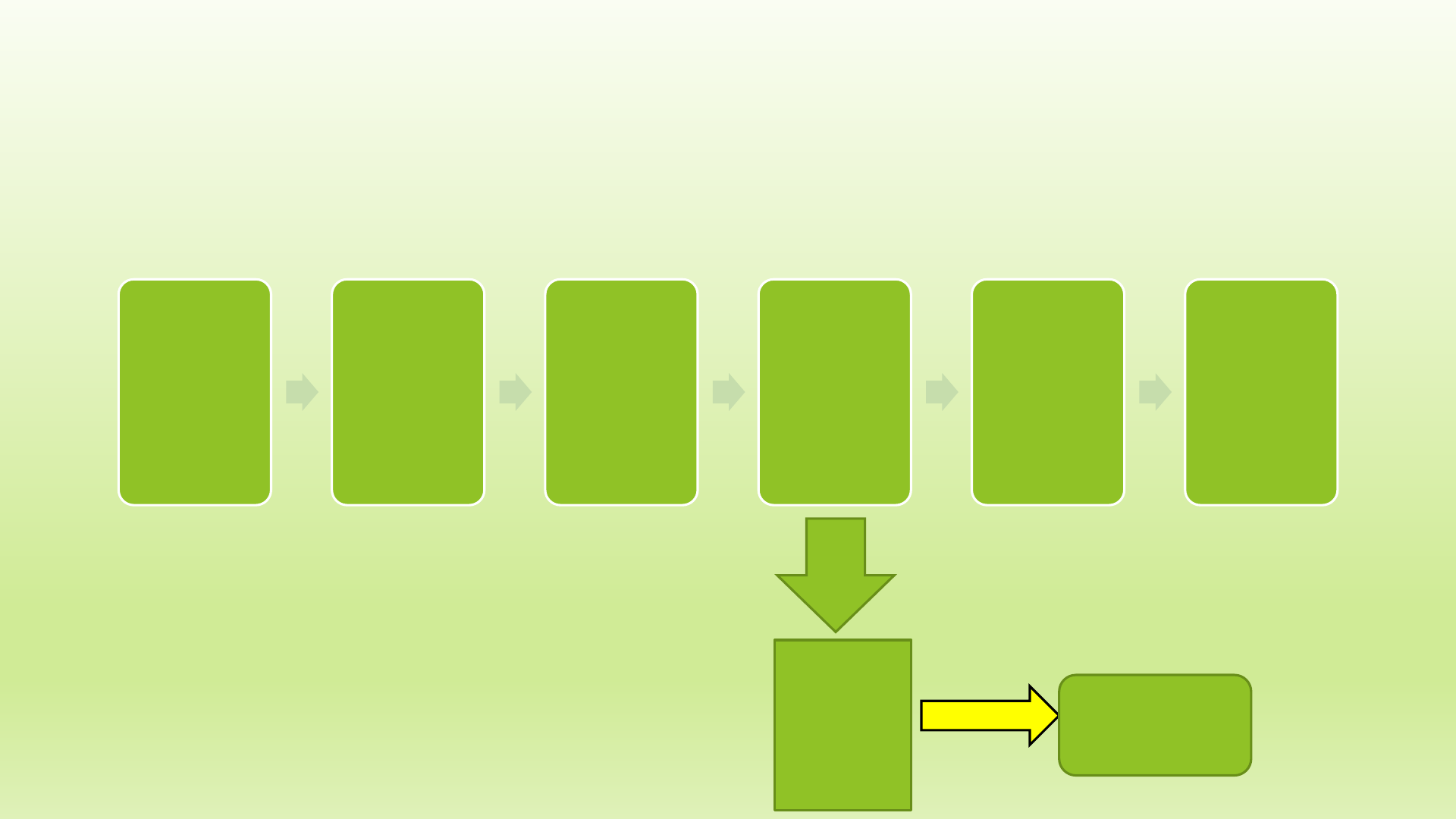
20-C License Process –
License
Received
License
Reviewed
GMP
Inspection
License
Issued
License
renewed
every 2
years
Facility
inspection
frequency
is based
on
product
risk
NOTE: A
processor
cannot offer
foods for sale
until they
have received
their license
Warning
Food found in the
market from an
‘unapproved’ is
subject to seizure

Good Manufacturing
Practices (GMPs)
GMP Category
GMP Requirement For:
Personnel
1. Disease control
2. Cleanliness
3. Education and training
4. Supervision of personnel with regards to these requirements
Building
and Grounds
1. Description of adequate maintenance of grounds/exterior of
facility
2. Facility/kitchen construction and design to facilitate sanitary
operations and maintenance
Sanitary Operations
1. Cleaning/sanitizing of physical facilities, utensils, and
equipment
2. Storage of cleaning and sanitizing substances
3. Pest control
4. Sanitation of food contact surfaces
5. Storage and handling of cleaned portable equipment and
utensils
Sanitary Facilities and
Controls
1. Water supply
2. Plumbing
3. Sewage disposal
4. Toilet facilities
5. Hand
-washing facilities
6. Rubbish and offal disposal
Equipment and Utensils
Design, construction, and maintenance of equipment and utensils
Processes and controls
Delineates processes and controls for:
1. Raw materials and other ingredients (separation, storage,
rotation,
etc)
2. Manufacturing operations (critical limits, allergen control,
cross contamination,
etc)
Warehousing
/ Storage
and distribution
Storage and transportation of final food product must protect
against contamination and deterioration of the food and its
container
Good manufacturing practices provide guidance
for manufacturing, testing, and quality assurance
of food to ensure that a food product is safe for
human consumption.
All GMP guidelines follow a few basic principles:
1. Instructions and procedures must be written
in clear and unambiguous language
2. Records produced during the
manufacturing/food process (temperature
controls, cleaning/sanitation schedules,
training, batch, lot codes, etc) must be
maintained
Provided in your
handout

GMP - Personnel
The facility management shall take all reasonable measures and precautions to assure the
following:
Disease control. No person affected by disease in a communicable form, or while a carrier
of such disease, or while affected with boils, sores, infected wounds, or other abnormal
sources of microbiological contamination, shall work in a food plant in any capacity in
which there is a reasonable possibility of food or food ingredients becoming contaminated
by such person, or of disease being transmitted by such person to other individuals.
Devise a policy
Share the policy with employees

GMP - Personnel
Cleanliness. all persons, while working in direct contact with food preparation, food
ingredients, or surfaces coming into contact therewith shall:
Wear clean outer garments, maintain a high degree of personal cleanliness, and conform to
hygienic practices while on duty, to the extent necessary to prevent contamination of food
products.
Wash their hands thoroughly (and sanitize if necessary to prevent contamination by undesirable
microorganism) in an adequate hand-washing facility before starting work, after each absence
from the work station, and at any other time when the hands may have become soiled or
contaminated.
Remove all insecure jewelry and, during periods where food is manipulated by hand, remove
from hands any jewelry that cannot be adequately sanitized.
If gloves are used in food handling, maintain them in an intact, clean, and sanitary condition.
Such gloves should be of an impermeable material except where their usage would be
inappropriate or incompatible with the work involved.
Wear hair nets, headbands, caps, or other effective hair restraints.
Not store clothing or other personal belongings, eat food or drink beverages, or use tobacco in
any form in areas where food or food ingredients are exposed or in areas used for washing
equipment or utensils.
Take any necessary precautions to prevent contamination of foods with microorganisms or
foreign substances including, but not limited to, perspiration, hair, cosmetics, tobacco,
chemicals and medications.

Personnel - The Good, the Bad and the Ugly
The Good The Bad The Ugly

Buildings and Grounds The Good, the Bad and the Ugly
The Good The Bad The Ugly

GMP – Sanitary Operations
You must have program in place to ‘maintain a clean and
sanitary operation’.
When possible a program must be available and documented
for the following:
➢ General maintenance of buildings and grounds
➢ Animal and vermin control
➢ Sanitation of equipment and utensils
➢ Sanitation/cleaning of walls, floors, ceilings etc

Sanitary Operations - The Good, the Bad and the Ugly
The Good The Bad The Ugly

GMP – Sanitary Facilities & Controls
Each facility shall be equipped with adequate sanitary facilities and
accommodations, including but not limited to the following:
Water supply
Sewage disposal
Plumbing
Toilet facilities
Hand-washing facilities
Rubbish and offal disposal

Sanitary Facilities & Controls The Good, he Bad and the Ugly
The Good The Bad The Ugly

GMP – Equipment & Utensils
All plant equipment and utensils should be:
suitable for their intended use,
so designed and of such material and
workmanship as to be adequately cleanable,
and
properly maintained.

Equipment The Good, the Bad and the Ugly
The Good The Bad The Ugly

GMP – Processes and Controls
Raw material and ingredients shall be segregated as necessary to
assure that they are clean, wholesome, and fit for processing
into human food
Raw materials shall be stored under conditions that will protect
against contamination and minimize deterioration.
Containers and carriers of raw ingredients should be inspected
on receipt and routinely to assure that their condition has not
contributed to the contamination or deterioration of the
products

GMP – Processes and Controls
When ice is used in contact with food products, it shall be made
from potable water
Food-processing areas and equipment used for processing human
food should not be used to process non-human food-grade
animal feed or inedible products unless there is no reasonable
possibility for the contamination of the human food.
Processing equipment shall be maintained in a sanitary condition
through frequent cleaning including sanitization where
indicated.
Insofar as necessary, equipment shall be taken apart for
thorough cleaning.

Processes and Controls - The Good, the Bad and the Ugly
The Good The Bad The Ugly

GMP – Warehouse, Storage and Distribution
All food packaging and storage, should be conducted under such conditions
and controls as are necessary to minimize the potential for undesirable
deterioration or contamination of the processed product or ingredients
Storage may require careful monitoring of such physical factors as:
Time,
Temperature,
Humidity,
Pressure,
Flow-rate
Processing operations as freezing, dehydration, heat processing, and refrigeration
Testing procedures shall be utilized where necessary to identify sanitation
failures or food contamination
Packaging processes and materials shall not transmit contaminants or
objectionable substances to the products

GMP – Warehouse, Storage and Distribution
Meaningful coding of products sold or otherwise distributed from
a manufacturing, processing, packing, or repacking activity
should be utilized to enable positive lot identification to
facilitate
Records should be retained for a period of time that exceeds the
shelf life of the product
Storage and transportation of finished products should be under
such conditions as will prevent contamination and will protect
against undesirable deterioration of the product and the
container

Warehouse, Storage and Distribution -The Good, the Bad and the Ugly
The Good The Bad The Ugly

Allergens
Food labels must identify the source of all major allergens in the food’s ingredient list
in simple language.
Unless the food source of a major food allergen is part of the ingredient’s common and
usual name (e.g. milk, wheat), it must be labeled in one of two ways. It may be added
in parenthesis after the ingredient (e.g. whey (milk), flour (wheat)) or can be
identified at the end of the ingredient list in a “contains” statement (e.g. contains
milk and wheat).
What are the major food allergens in the US? Worldwide?
Milk,
Eggs,
Fish,
Crustacean shellfish (e.g. shrimp, crab, lobster),
Tree nuts (e.g. almonds, walnuts, pecans, etc.),
Peanuts,
Wheat,
Soybeans.

Allergens - The Good, the Bad and the Ugly
The BadThe Good The Ugly

Cross Contact/Contamination
What is cross-contact/contamination?
Cross-contact occurs when a residue or trace amount of an
allergen unintentionally crosses over into a product that
doesn’t have that allergen.
Cross-contact (the inadvertent introduction of an allergen into
a product) is generally the result of environmental exposure
during food processing or handling.
Cross-contact occurs when:
multiple foods are manufactured on the same processing line,
through the misuse of rework,
as a result of ineffective cleaning, or
from the generation of significant dust containing the allergen.

Cross Contact/Contamination
How can cross-contact be prevented?
Cross-contact cannot always be prevented. However, by developing and implementing an
Allergen Control Plan, one can either prevent or at least minimize allergen cross-contact
as much as possible.
The Allergen Control Plan is a written document outlining controls put in place
regarding the:
storage, handling, and processing of allergens and
the identification of places where cross-contact is likely to occur.
NOTE: Prevention and monitoring methods are included to prevent cross-contact.
An effective Allergen Control Plan should start with the production of raw materials,
storage and handling of raw materials, and every step in the manufacturing process
through the packaging and labeling of the finished product.
The critical points where allergens may be introduced into the product during
manufacturing should be identified and a system established to monitor these points
to ensure unintentional cross contact is prevented.

Cross Contact/Contamination
What is precautionary allergen labeling?
Precautionary allergen labeling (sometimes also referred
to as allergen advisory labeling) is a voluntary warning to
consumers (e.g. may contain milk) added after the
ingredient list.
It’s goal is to indicate a product not intended to contain
a specific allergen(s) may sporadically contain that
allergen due to unintentional and unavoidable cross-
contact in the manufacturing process even after
implementing a comprehensive Allergen Control Plan.

Traceability - Recall Plan
Traceability
Ability to track food through
the food system back to their
source and forward to the next
destination
DOES NOT prevent a foodborne
outbreak
CAN quickly identify the source
of a product and speed an
investigation
CAN limit damage to the
consumer
CAN prevent damage to the
innocent ‘processor’
Product Recall
Recalls are actions taken by a
firm to remove a product from
the market.
They may be conducted
on your own initiative,
By request of your buyer,
by NYSDAM/FDA request, or
by FDA order under court
order.

Recall Plan Development
How to make your products traceable
Keep records of
name of the product,
the ingredients & manufacturers,
the processing area,
processing dates (or group of dates),
LOT numbers, and
where it was sent.
Use processing records, receipts, transportation bills, and
storage records to provide documentation.
Keep records for products that are held in storage before
distribution
NOTE: ALWAYS
CHECK THAT IT
WORKS

Preparing for your ‘Inspection’ through a
‘Self-Audit’

Personnel
Grounds/
Building
Sanitation
Equipment
Storage/
Transportation
Food Labeling
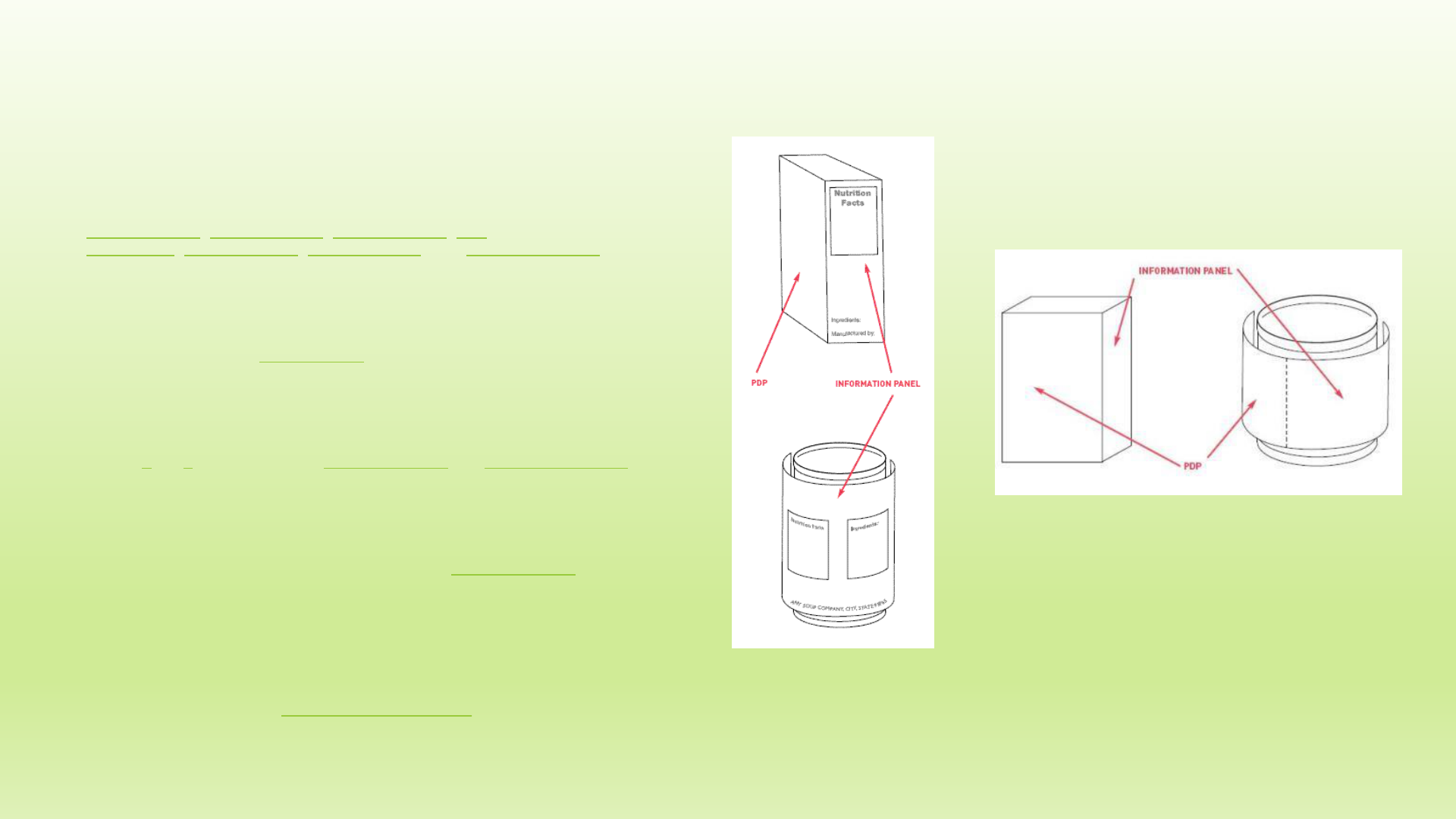
1. Where should label statements be placed on containers and packages?
Answer: There are two ways to label packages and containers:
a. Place all required label statements on the front label panel (the
principal display panel or PDP), or,
b. Place certain specified label statements on the PDP and other labeling
on the information panel (the label panel immediately to the right of the
PDP, as seen by the consumer facing the product).
21 CFR 101.1, 21 CFR 101.2, 21 CFR 101.3, 21_
CFR 101.4, 21 CFR 101.5, 21 CFR 101.9, and 21 CFR 101.105
2. What are the PDP and the alternate PDP?
Answer: The PDP, is that portion of the package label that is most likely to be
seen by the consumer at the time of purchase. Many containers are designed
with two or more different surfaces that are suitable for display as the PDP.
These are alternate PDPs. 21 CFR 101.1
3. What label statements must appear on the PDP?
Answer: Place the statement of identity, or name of the food, and the net
quantity statement, or amount of product, on the PDP and on the alternate
PDP. The required type size and prominence are discussed
in sections 4and 5 of this guidance. 21 CFR 101.3(a) and 21 CFR 101.105(a).
4. Which label panel is the information panel?
Answer: The information panel is the label panel immediately to the right of
the PDP, as displayed to the consumer. If this panel is not usable, due to
package design and construction, (e.g., folded flaps), then the information
panel is the next label panel immediately to the right. 21 CFR 101.2(a)
5. What is information panel labeling?
Answer: The phrase "information panel labeling" refers to the label
statements that are generally required to be placed together, without any
intervening material, on the information panel, if such labeling does not appear
on the PDP. These label statements include the name and address of the
manufacturer, packer or distributor, the ingredient list, nutrition labeling and
any required allergy labeling. 21 CFR 101.2(b) and (d), Section 403(w) of the
FD&C Act

6. What type size, prominence and conspicuousness is required?
Answer: For information panel labeling, use a print or type size that is prominent, conspicuous and
easy to read. Use letters that are at least one-sixteenth (1/16) inch in height based on the lower case
letter "o". The letters must not be more than three times as high as they are wide, and the lettering
must contrast sufficiently with the background so as to be easy to read. Do not crowd required
labeling with artwork or non-required labeling.
Smaller type sizes may be used for information panel labeling on very small food packages as
discussed in 21 CFR 101.2(c) & (f).
Different type sizes are specified for the Nutrition Facts label. (see section 7)
The type size requirements for the statement of identity and the net quantity statement are discussed
in sections 4 and 5 of this guidance. 21 CFR 101.2(c)
7. What is the prohibition against intervening material?
Answer: Information that is not required by FDA is considered intervening material and is not
permitted to be placed between the required labeling on the information panel (e.g., the UPC bar
code is not FDA required labeling). 21 CFR 101.2(e)
8. What name and address must be listed on the label?
Answer: Food labels must list:
a. Name and address of the manufacturer, packer or distributor. Unless the name given is the
actual manufacturer, it must be accompanied by a qualifying phrase which states the firm's
relation to the product (e.g., "manufactured for "or "distributed by");
b. Street address if the firm name and address are not listed in a current city directory or
telephone book;
c. City or town;
d. State (or country, if outside the United States); and
e. ZIP code (or mailing code used in countries other than the United States). 21 CFR 101.5
Food Labeling – Continued

HAZELS
Blueberry Muffin Mix
Artificially flavored
NET WT. 12 oz. (340g)
HAZELS
Wild Blueberry
Mix
Naturally flavored
SO GOOD
NET WT. 12oz
.
Enriched Bleached flour (niacin, iron, riboflavin),
sugar, artificial blueberry bites (palm oil, corn
flour, artificial flavor, red coloring), vegetable oil,
baking soda, corn starch, salt, eggs, sodium
silicoaluminate.
May contain: milk and soy
Serving Size: 1/6 pouch (31g), Servings per Container: 6
Amount Per Serving, Calories 120,Calories from Fat 25, %
Daily Value, Total Fat 2.5g (4%) , Saturated Fat 1g (4%),
Trans Fat, 1g,Cholesterol 0mg (0%),Sodium 230mg
(10%),Total Carb 23g (8%)
Sugars 11g, Protein 1g, Iron 2%, Thiamin 4%
Riboflavin 2%, Niacin 2%,
Not a significant source of dietary fiber, vitamin A and vitamin
C and calcium.
* Percent Daily Values are based on a 2,000 calorie diet.
Distributed by Lazurne’s LLC
Hamilton, NY
Hazelsfoods.com
Enriched Bleached flour (wheat flour, niacin,
iron, riboflavin), sugar, artificial blueberry
bites (palm oil, corn flour, artificial flavor, red
40, blue 1), vegetable oil (sunflower, and
peanut oil), baking soda, corn starch, salt,
eggs, sodium silicoaluminate (anticaking
agent).
Distributed by Lazurne’s LLC
64 Lake Rd.
Hamilton, NY 12202
A SPOT THE DIFFERENCE B
Take a look at the label and circle what you
think is wrong with the picture!

Food Safety Training
As required by Agriculture and Markets Law §251-z-312, the applicant for a food processing
establishment license shall furnish evidence of his or her experience and competency to
operate the establishment. One can demonstrate such competency by successfully
completing a food safety course approved by the Department.
The food safety course shall consist of not less than eight hours of training received
within two years prior to the commencement of the license period or, if the program
required the passing of a test, within five years prior to the commencement of the
license period.
The program shall cover one or more of the following topics:
New York State food safety statutes and regulations;
Food microbiology, including a review of pathogenic and spoilage microorganisms;
food-borne illnesses, including causative agents, symptoms, and prevention;
HACCP (Hazardous Analysis Critical Control Point);
Cleaning and sanitation;
Personal hygiene;
Temperature control, including heating, cooling, and storage standards; and
Food security, including identifying risks, implementing preventive measures and pest
control.

‘Scheduled’ Process Review
1 NYCRR 271.9.3:
A scheduled process or process review must be conducted on food products that
ordinarily do not have a processing step that addresses the rationale for how the
potential public health hazards and insanitary conditions addressed by the
relevant regulatory sections will be alternatively addressed by the new process.
The schedule process or process review must outline at a minimum recipe
testing/formulation, critical control points (to avoid contamination and control
hazards), processing steps, storage requirements, distribution and selling
conditions/restrictions, etc.
A scheduled process or process review of your new product can be obtained by
contacting any of the parties listed under the ‘Process Review
1
’ section on the
website
1
Listing of ‘Recognized Processing Authority’ is available for download from our website

‘Scheduled’ Process Review
The department requirements for producing a for wholesale distribution are as
follows:
Completion of a scheduled hazard/process review conducted by a recognized process
authority
1
.
Completion of Article 20-C Food processing establishment license application.
Compliance with 1NYCRR Part 261 – Current good manufacturing practices.
Compliance with 1NYCRR Part 59.1 – Packaging and labeling of food (including
statement of identify, net quantity statements, ingredients, warnings, refrigeration
statements etc) See labeling slides
The department requirements for producing a product for retail sale are as follows:
Adherence to a variance as required under 1NYCRR Part 271.9 – Retail food store
sanitation regulations, compliance and enforcement.
Completion of Article 20-C Food Processing Establishment license application.
Compliance with 1NYCRR Part 271 - Retail food store sanitation regulations.
Compliance with 1NYCRR Part 59.1 – Packaging and labeling of food (including warning
refrigeration statements etc).
1
Listing of ‘Recognized Processing Authority’ is available for download from our website

Helpful Resources for Small Scale Food
Processors
Request the publication: Small Scale Food Entrepreneurship: A Technical
Guide for Food Ventures from Elizabeth Keller, 315-787-2273
or [email protected] or access the online
version: http://www4.gsb.columbia.edu/filemgr?file_id=738927
Product development, processing and distribution assistance is also available
from Nelson Farms at SUNY Morrisville – 315-655-8831
or www.nelsonfarms.org
To learn about small scale food processing activities in NYS, join the NYS
Small Scale Food Processors Association: www.nyssfpa.com and become a
member of Pride of NY – 800-554-4501

NYSDAM – Excellent Food Safety Resources
Review the following Circulars on the NYSDAM
website:
Circular 951 - Pursuant to the Licensing of
Food Processing Establishments
Circular 938 – Rules and Regulations Relating
to Food Processing Establishments
Circular 933 - Good Manufacturing Practices
BE READY FOR YOUR NEXT NYSDAM INSPECTION BY CHECKING
OUT OUR INSPECTION CHECKLIST/CATEGORIES

Regional Offices of the NYS Department of
Agriculture and Markets
Division of Food Safety and Inspection
55 Hanson Place,
Brooklyn, NY 11217-1583
Phone: 718-722-2876
Division of Food Safety and Inspection
Electric Tower Building, 535 Washington
Street, 2nd Floor Suite 203,
Buffalo, NY 14203
Phone: 716-847-3185
Division of Food Safety and Inspection
900 Jefferson Road, Rochester, NY 14623
Phone: 585-487-0200
Division of Food Safety and Inspection
NYS Fairgrounds – Art and Home Center, Syracuse, NY 13209
315-487-0852
US FDA
300 Pearl Street, Suite 100, Buffalo, NY 14202
716-551-4478
US FDA New York District Office
158-15 Liberty Avenue, Jamaica, NY 11433
718-662-5447
United States Department of Agriculture – Food Safety
Inspection Service (USDA FSIS)
5 Washington Square, Albany, NY 12205
518-457-4492

Focus on Prevention
FDA Food Safety
Modernization Act (FSMA)

Why is the law needed?
Foodborne illness is a significant burden
About 48 million (1 in 6 Americans) get sick each year
128,000 are hospitalized
3,000 die
Food supply more high-tech and complex
15 percent of U.S. food supply is imported
More foods in the marketplace
New hazards in foods not previously seen

Main Themes of the Legislation
Prevention
Inspections,
Compliance,
and Response
Import Safety
Enhanced
Partnerships

General Approach to FSMA
5. Review
& Adjust
2. Understand
Cause
3. Implement
Preventive
Controls
4. Monitor
Effectiveness
FSMA Preventive Controls
Comprehensive preventive controls
for human and animal food
facilities
Prevention is not new, but Congress
has given FDA explicit authority to
use the tool more broadly
Produce safety standards
Supplier verification standards
Import standards
Transportation standards
1. Identify Hazard

HACCP
HACCP (Hazard Analysis & Critical Control Points) Plans are mandated by FDA
regulations for certain products and processes, specifying procedures to be followed
to minimize contamination and to minimize/eliminate chemical, physical and
biological hazards when processing foods.
HACCP plans are required for wholesale sale (not for retail) of:
Seafood,
Dairy,
Meat and poultry products,
Juice and cider processing facilities
Registration with FDA? Processors and food facilities are required
to register with the FDA according to the Bioterrorism Act of 2002.
Registration information may be found at:
https://www.fda.gov/Food/GuidanceRegulation/FoodFacilityRegist
ration/default.htm

HACCP -vs- FSPC
16
HACCP
Built on a foundation of GMP’s
and prerequisite programs
Focuses on specific Process
Control Steps
Hazard Analysis
HACCP Plan – All CCP’s have critical
limits.
Monitoring, Corrective
Actions, Verification, Record
Keeping
What you will do
Food Safety Preventive
Controls
(aka FSMA)
Built on a foundation of GMP’s
and prerequisite programs
HACCP on Steroids - Includes other
elements in addition to process control steps
Hazard Analysis – Includes radiological
hazards
Food Safety Plan – not all preventive
controls have critical limits.
Monitoring, Corrective Actions,
Verification, Record Keeping
How you will do it

Components of Preventive Controls for
Human Foods Rule
17
Key Requirements:
Establish a Food Safety System that includes an analysis of
hazards and risk based preventive controls leading to the
creation of the Food Safety Plan…
▪ Hazard Analysis
▪ Preventive Controls
▪ Oversight
- Monitoring
- Corrective Action & Corrections
- Verification

Contents of a Food Safety Plan
Process
Control
Sanitation
Control
Allergen
Control
Supply
Chain
Program
Hazard
Analysis
Recall
Plan
Food Safety Plan
Including procedures for monitoring,
corrective action and verification
GMP’s and Other
Prerequisite Programs

Modified Requirements for Qualified Facilities
(Section 103)
Facility is exempt if:
Defined as very small business;
OR
The facility has a limited annual monetary value of
sales
Limited annual monetary value of sales is defined as:
During the last 3 years, sales were less than
$500,000;
AND
Sales to "Qualified End Users" exceed sales to others

Examples of Compliance with
Prevention Standards
Current Good Manufacturing Practices (cGMPs)
Sanitation
Training for supervisors and employees
Hazard Analysis and Risk-Based Preventative
Controls
Food Safety Plan
Hazard analysis
Preventive controls (chemical, physical,
biological)
Control monitoring, verification, validation
Recall contingency plan
Supplier verification activities
Will FSMA
apply to
YOU?
Check out
our website
for further
details

Transportation of Food
Sec. 111. Sanitary Transportation of Food
Addresses implementation of the Sanitary Food Transportation Act of 2005, which requires
persons engaged in food transportation to use sanitary transportation practices to ensure
that food is not transported under conditions that may render it adulterated.
Specific exemptions to this include:
Transportation of Grade “A” milk and milk products
Food establishments authorized by the regulatory authority to operate when engaged as
receivers, or as shippers and carriers in operations in which food is delivered directly to
consumers, or to other locations the establishments or affiliates operate that serve or sell
food directly to consumers. (Examples include restaurants, supermarkets and home grocery
delivery services.)
Businesses transporting molluscan shellfish (such as oysters, clams, mussels or scallops) that
are certified and inspected under the requirements established by the Interstate Shellfish
Sanitation Conference’s (ISSC) National Shellfish Sanitation Program (NSSP) and that
transport the shellfish in vehicles permitted under ISSC authority.

Import Prevention Program
Sec. 301 Foreign Supplier Verification Program (FSVP)
Requires importers to conduct risk-based foreign supplier verification activities
to verify that food imported into the United States is not adulterated and that
it was produced in compliance with FDA’s preventive controls requirements
and produce safety standards
Sec. 307. Third Party Auditor Accreditation
Can be used by importers for supplier verification under FSVP
The following third party certifying bodies specialize in food safety inspection
and sanitation (please be advised, however, that NYSDAM makes no
representation as to the competency of any particular certifying body nor
should or will it be held liable for any action taken by a certifying body:
http://www.sqfi.com/
http://www.mygfsi.com/
http://www.brcglobalstandards.com/
http://www.primuslabs.com/services/AuditorTypes.aspx

FDA and NYSDAM FSMA Implementation Approach
NYSDAM Implementation (multiple year
approach)
Conduct outreach to determine Industry
size, exemptions, applicability
Develop a dataset of ‘affected’ facilities
Classify facilities : Facilities will be ranked
according to their risk classification and
prioritized based on public health
protection
Educate those affected
Engage stakeholders to help determine
reasonable and practical ways to implement
preventive control provisions
Work with stakeholders to implement
provisions
Visit and work with affected facilities (large,
small, very small) to identify gaps with
compliance and educate accordingly
Begin inspection protocol
Begin enforcement protocol
FDA Implementation
Items are prioritized based on public
health protection
Engage with stakeholders to help
determine reasonable and practical ways
to implement preventive control provisions
Implementation progress at
http://www.fda.gov/fsma
Registration with the FDA will
be key!
NOTE: FDA registration does
not substitute the need to
notify NYSDAM of your
business or completion of
license application

Questions?
Don’t forget to pick
up all your resource
guides/handouts!!
